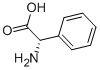
L-Phenylglycine: Fermentative Production & Synthetic Optimization
L-phenylglycine is an important non-protein amino acid derivative. It exists as a white crystalline powder at ambient temperature, with a melting point exceeding 300°C. It exhibits chiral properties and serves as a key chiral auxiliary in asymmetric synthesis. Its preparation can be achieved via the Strecker synthesis of benzaldehyde or the reductive amination of glyoxal. Its most extensive applications lie within the pharmaceutical sector, serving as a core raw material for synthesising antibiotics such as ampicillin and cefazolin. L-phenylglycine is also utilised in the production of agrochemicals, food additives, and cosmetics. In biochemical research, this substance facilitates investigations into protein interactions, establishing itself as an indispensable organic synthesis intermediate across multiple fields.

Fermentative production of phenylglycines
To date, more than 900 naturally occurring amino acids have been identified (Lu and Freeland 2006) of which the 20 proteinogenic L-amino acids only constitute 2%. The majority of the residual 98% of non-proteinogenic amino acids serve as building blocks for bioactive natural compounds. S. pristinaespiralis is the producer of the streptogramin antibiotic pristinamycin, which consists of the two chemically non-related substances pristinamycin I (PI) and pristinamycin II (PII). PI is synthesized by the nonribosomal peptide synthetases (NRPSs) SnbA, SnbC, and SnbDE, whereby the latter one incorporates L-phenylglycine as the final amino acid into the growing PI peptide chain. L-phenylglycine in S. pristinaespiralis is suggested to originate from the shikimate pathway. As a first metabolic step, phenylpyruvate is converted to phenylacetyl-CoA by the action of a pyruvate dehydrogenase–like complex PglB/C. Phenylacetyl-CoA is oxidized to benzoylformyl-CoA via the Phg dioxygenase PglA. The CoA residue from benzoylformyl-CoA is cleaved off by the thioesterase PglD, resulting in the formation of phenylglyoxylate. In a final reaction step, phenylglyoxylate is converted to L-phenylglycine by the aminotransferase PglE. As PglE uses L-phenylalanine as amino group donor for the transamination reaction, phenylpyruvate is formed as the α-keto acid product, which can re-enter Phg biosynthesis as a precursor. In this study, we describe the development of a synthetic biology-derived D-Phg pathway. Furthermore, we report on genetic engineering approaches in order to optimize Phg production in actinomycetal expression strains.[1]
To obtain constructs for the fermentative production of L-phenylglycine, the native ~ 6-kb lpg operon from S. pristinaespiralis was cloned into the integrative vector pRM4 under control of the constitutive ermE promoter, resulting in the expression construct pYM/lpg. For production of the D-Phg enantiomer, an artificial D-Phg operon (dpg) was generated on the basis of the native lpg operon from S. pristinaespiralis: In a synthetic biology approach, the gene pglE, encoding the L-phenylglycine aminotransferase in S. pristinaespiralis, was exchanged by the gene hpgAT from P. putida, which codes for a stereospecific D-Phg aminotransferase. This D-Phg aminotransferase is the only currently known L to D stereo-inverting aminotransferase. A logic strategy to further increase Phg production is to perform a combinatory genetic engineering approach and inactivate all the above-mentioned genes in S. pristinaespiralis. An additional target gene for further production improvement is papR2, which encodes a SARP-type transcriptional regulator that is suggested to activate the L-phenylglycine operon in S. pristinaespiralis. One could assume that this pathway is already evolutionary optimized and under appropriate conditions might be more powerful than an artificially assembled pathway harboring genes from different origins. However, this would require further investigations. Overall, the Phg operons represent promising biobricks for Phg-related production processes. A purely fermentative production route has mainly been prevented by the absence of a natural Phg pathway. In this study, we could describe the functionality of the natural L-phenylglycine operon from S. pristinaespiralis and its derived D-Phg operon obtained by a synthetic biology approach. The new fermentative Phg production route serves as a basis to replace the environmentally unfriendly industrial Phg production process.
Improved L-phenylglycine synthesis
L-phenylglycine (L-phg) is a non-proteinogenic amino acid, which is used as the building block of virginiamycin S, pristinamycin I, penicillin, and antitumor compound taxol. Currently, the synthesis of L-phg is based on petrochemical feedstocks. The process uses environmentally harmful organic solvents and poisonous reagents and the low enantioselectivity which does not meet the demands of environmental safety and green chemistry. A fermentative process to produce chiral amino acids is preferable because of its high enantioselectivity, sustainable raw material, and relatively reduced environmental detriments. Previous studies have reported overproduction of L-phenylglycine based on glucose, l-phenylalanine (L-phe), and other substrates. In the meantime, researchers have developed other biological routes for L-phg synthesis. In one, racemic mandelate is used as the substrate. Mandelate racemase (MR) coupled with d-mandelate dehydrogenase (MD) catalyzes the substrate to produce PG, which is converted to L- or D-phg by HpgAT or DAT. A recent example for L-phg production used leucine dehydrogenase (LeuDH) to catalyze the conversion of PG to L-phg using NADH and NH4+ as cofactor. To get sufficient redox supply, formate dehydrogenase was co-expressed, which regenerated NADH by oxidizing formate to CO2. By co-expressing of FDH with LeuDH, 60.2 g/L L-phg was obtained with 99% yield, which showed a satisfactory productivity and enantioselectivity towards L-phenylglycine. In this study, we exemplified a self-sufficient biotransformation system from amino acid L-phe to its value-added chiral amine derivatives L-phg.[2]
L-phg is an important building block of antibiotics, antitumor drug taxol, drugs used to treat Alzheimer's and many other diseases. Many progresses have been made on synthesis of L-phg in a biological way. In this study, we built a L-phenylglycine synthesis pathway in E. coli. However, only a small amount of L-phe was utilized to synthesis L-phg. As study showed that enzyme activities of HmaS and Hmo from S. coelicolor were 0.501 and 1.822 U/mg. At the same time, the solubility of these two enzymes in E. coli were low. Thus, HmaS and Hmo was considered as the major bottleneck for L-phenylglycine production. To retain more NADH, an orthogonal interaction pair slig-SH3 was applied in order to colocalize LeuDH and GDH. This protein scaffold strategy was first used for mevalonate biosynthesis. By altering the stoichiometry of the SH3, PDZ, and GBD domains, three mevalonate biosynthetic enzymes were arranged in a designable manner, which lowered the host-expressing burden and increased the production of mevalonate 77-fold. Protein scaffolds have been applied to incorporate sequential enzymes in gamma-aminobutyric acid, polyhydroxybutyrate, and resveratrol production. In this work, a scaffold was built between the synthetic pathway and cofactor regeneration module. In this way, cofactor NADH, and co-substrate NH4+ generated by GDH could be directly utilized by LeuDH. Thus, L-phg increased by 2.1-fold compared with no scaffold. The final L-phenylglycine level reached 3.72 mM, which was the highest L-phg titer from L-phe or glucose.
References
[1]Moosmann D, Mokeev V, Kulik A, Osipenkov N, Kocadinc S, Ort-Winklbauer R, Handel F, Hennrich O, Youn JW, Sprenger GA, Mast Y. Genetic engineering approaches for the fermentative production of phenylglycines. Appl Microbiol Biotechnol. 2020 Apr;104(8):3433-3444. doi: 10.1007/s00253-020-10447-9. Epub 2020 Feb 20. PMID: 32078019; PMCID: PMC7089894.
[2]Wang, P., Zhang, X., Tao, Y., Lv, X., Cheng, S., & Liu, C. (2022). Improved L-phenylglycine synthesis by introducing an engineered cofactor self-sufficient system. Synthetic and Systems Biotechnology, 7(1), 513–521.
Lastest Price from L-Phenylglycine manufacturers

US $0.00-0.00/kg2026-01-19
- CAS:
- 2935-35-5
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20MT

US $0.00/Kg/Drum2025-04-21
- CAS:
- 2935-35-5
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 500kgs